Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Clinical trial supply chains are essential for ensuring investigational drugs reach patients safely and on time. Careful planning, maintaining adequate stock, and flexible trial designs help reduce delays. Technology, including tracking systems, data analytics, and blockchain, improves efficiency and ensures accuracy of data. Following international regulations carefully prevents errors and interruptions, making trials more reliable and helping sponsors manage costs effectively.

Clinical trials are a high-cost stage in the development of new medicines. The clinical trial supply chain refers to the planning and management of supplying investigational drugs to different trial sites for participant use.1,2 The main objective is to ensure adequate availability of drugs while minimising operational costs.
Due to uncertainty in patient enrolment and treatment completion, a certain level of safety stock is maintained, which increases inventory levels. In addition, unused drug supplies must be disposed of as per regulatory requirements, adding to the overall cost.1 Effective management also includes timely distribution, quality control, and accountability.2
The clinical trial supply chain follows a systematic process from manufacturing to patient use, ensuring that investigational drugs reach clinical sites efficiently. It involves multiple stages, including production, processing, packaging, and distribution before administration to trial participants.
The total time taken to move drugs from manufacturing to distribution centres may be up to 1 year or even longer.3 Figure 1 shows the movement of investigational drugs from manufacturing to patient administration stages.
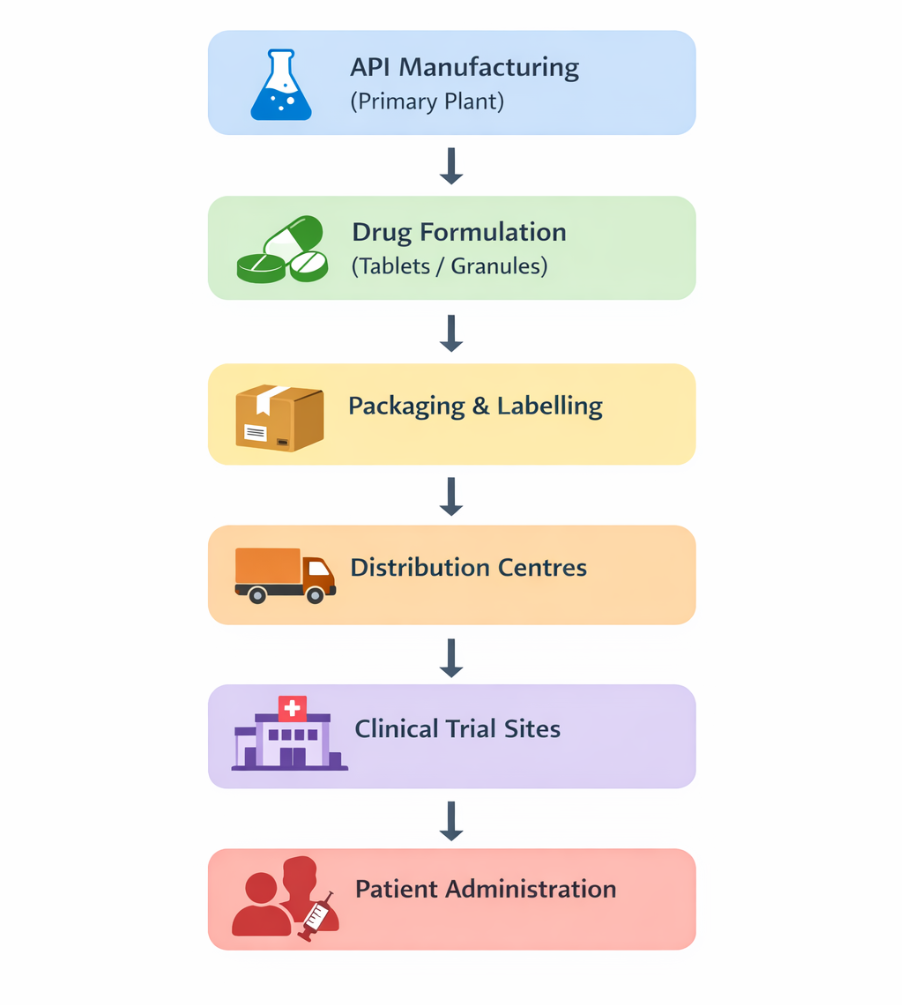
Clinical trial supply chains are different from conventional supply systems because they deal with drugs that are still under development.3 These systems are complex due to uncertain demand, long timelines, and global challenges.4 Common clinical trial supply chain issues include:
Clinical trial drugs take a long time to produce, sometimes up to one year or more. It is also costly and difficult to make new batches quickly when needed.
The demand for drugs changes over time due to patient enrolment and dropout. This makes planning difficult and may lead to an imbalance in supply.
Shortage of drugs can occur because demand is not predictable. These drugs cannot be easily replaced, which may affect patient treatment.
Some patients may stop treatment due to lack of improvement or loss of interest. New patients must be recruited, increasing drug demand.
Costs include stock-out, relocation, and storage costs at clinics. Managing these costs makes the system more complex.
Transporting drugs between locations requires time and incurs cost. This reduces the efficiency of redistribution methods.3
Different countries have different rules for trials, labelling, and data protection. Logistics issues like transport delays and customs can also affect supply.4
Clinical trial supply chains can be managed more effectively by focusing on proper planning and preventive strategies to avoid disruptions.4 Using flexible approaches and better coordination can help maintain smooth supply and continuous trial progress.3 Strategies to reduce clinical study supply chain risk include:
Clinical trial sponsors should prepare backup plans to manage unexpected disruptions, such as delays in production, shipment, and site activities.
Using flexible trial designs allows changes to be made during disruptions. This helps trials continue even when supply chain issues occur.
Remote monitoring and virtual platforms allow patients to participate without travelling. This reduces dependence on physical site visits during disruptions.4
Proper planning of inventory between distribution centres and clinical sites is important. Techniques like replenishment and redistribution help prevent shortages.3
Using central hubs helps manage and distribute supplies efficiently across regions. This reduces delays and improves control over shipments.
Understanding and following the regulations of different countries helps avoid delays. Experts can help ensure proper handling, labelling, and documentation.
Regular monitoring of supply chain activities helps identify risks early. Coordination between teams ensures smooth and timely delivery of trial materials.4
The table below shows key challenges in clinical trial supply chains and simple strategies to reduce disruptions effectively.
| Clinical Trial Supply Chain Issues | Strategies to Reduce Disruptions |
| Long production time for trial drugs | Proper planning and having backup plans can help manage delays |
| Uncertain demand due to patient enrolment changes | Using data and flexible trial designs can help adjust supply as needed |
| Risk of stock-outs at clinical sites | Keeping safety stock and planning inventory properly can prevent shortages |
| High inventory and storage costs | Better stock management and using central hubs can reduce costs |
| Global regulatory and logistics issues | Following regulations carefully and improving coordination across regions can avoid delays |
Technology helps improve clinical trial supply chains by making processes faster, more accurate, and better coordinated.4 It supports efficient management of inventory, data, and communication across multiple trial sites.5
Advanced tracking tools and cloud platforms help monitor drug movement and stock levels. This reduces delays and prevents shortages or excess supply.4
Data analytics and artificial intelligence help predict demand and optimise inventory. This improves planning and reduces wastage and costs.
Blockchain technology ensures data is secure and cannot be changed. It improves transparency and trust in clinical trial data.4
Clinical trial supply chains play an important role in ensuring that drugs reach patients on time and trials run smoothly. By managing challenges, planning appropriately, and adopting new technologies, supply chain disruptions can be reduced. A well-managed system helps in improving efficiency, controlling costs, and ensuring better outcomes in clinical research, making the overall trial process more reliable and effective.
1. Chen Y, Pekny JF, Reklaitis GV. Integrated Planning and Optimization of Clinical Trial Supply Chain System with Risk Pooling. Ind Eng Chem Res. Published online August 14, 2012:120814121202002. doi:10.1021/ie300823b
2. Supply chain planning for clinical trials : a practical guide in SearchWorks catalog. Accessed March 23, 2026. https://searchworks.stanford.edu/view/in00000183469
3. A Study on the Optimal Inventory Allocation for Clinical Trial Supply Chains. Applied Mathematical Modelling. 2021;98:161-184. doi:10.1016/j.apm.2021.04.029
4. (PDF) THE FUTURE OF CLINICAL TRIALS: ETHICAL, TECHNOLOGICAL, AND SUPPLY CHAIN PERSPECTIVES. ResearchGate. September 22, 2025. doi:10.56726/IRJMETS67432
5. Abdullahi HB, Ado AP. Secure Data Analytics for Clinical Trial Supply Chains. International Journal of Engineering Research. 2024;13(02).