Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124

A Clinical Research Organization (CRO) is a company that provides outsourced research services to pharmaceutical, biotechnology, and medical device companies. CROs help plan, manage, monitor, and analyze clinical trials required to prove that a drug or device is safe and effective.
A CRO helps bring new medicines and medical innovations from the laboratory to real patients through structured and compliant clinical trials.
Clinical trials are complex, regulated, and expensive. Sponsors (pharma or biotech companies) partner with CROs because they:
Without CROs, many companies would struggle to manage multi-center, multi-country clinical trials efficiently.
In the medical field, CRO stands for either Clinical Research Organization or Contract Research Organization — a company hired on a contractual basis to support clinical research and drug development for pharmaceutical, biotechnology, and medical device companies. CROs provide specialized services such as clinical trial planning and management, patient recruitment, data collection and analysis, regulatory compliance support, and reporting, helping sponsors run research efficiently without maintaining large in-house teams.
Cliniexperts Research operates as a dedicated clinical research partner, supporting pharmaceutical, biotech, and medical device companies with high-quality and compliant trial management solutions.
Here’s how Cliniexperts Research adds value:
Cliniexperts Research assists sponsors with:
This ensures smooth approvals and reduced regulatory delays.
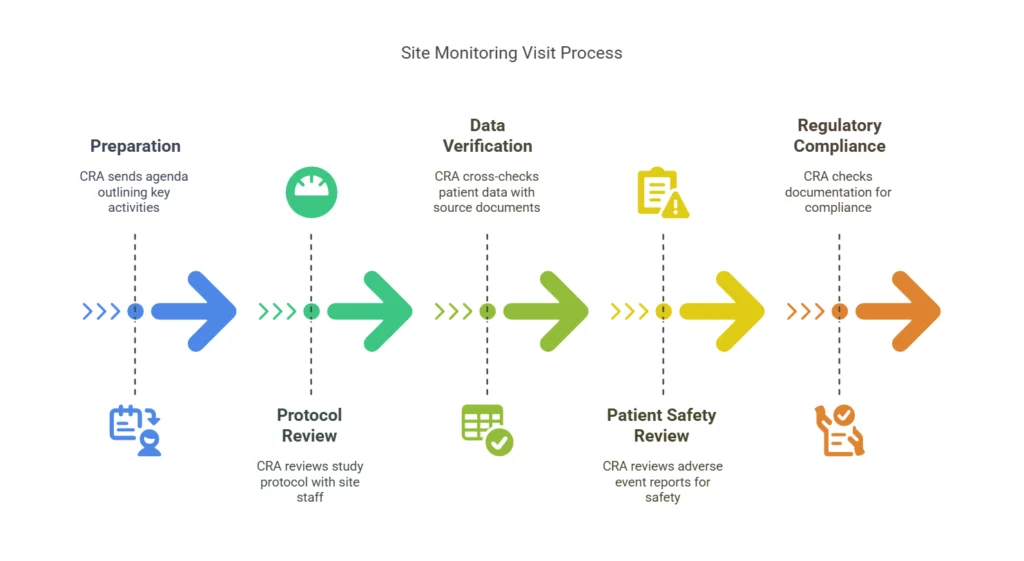
The team provides:
Their structured monitoring approach ensures data accuracy and patient safety.
Accurate data is the backbone of any clinical trial. Cliniexperts Research focuses on:
This improves trial credibility and regulatory acceptance.
For emerging biotech companies and medical device startups, managing in-house clinical teams can be expensive. Cliniexperts Research provides:
This helps sponsors optimize operational costs while maintaining quality.
From feasibility to final reporting, Cliniexperts Research supports:
Their integrated approach ensures seamless communication between sponsors, investigators, and regulatory authorities.
CROs essentially act as clinical research service providers on a contract basis that manage various aspects of clinical trials and research activities. They reduce costs, accelerate development timelines, and bring expertise in areas like biostatistics, monitoring, and regulatory affairs. CROs can be full-service (handling entire trial lifecycles) or specialized/niche (focused on certain phases or therapeutic areas).
In the medical field, a CRO (Clinical Research Organization) plays a crucial role in transforming scientific research into approved medical treatments. By managing clinical trials, ensuring compliance, and maintaining data integrity, CROs bridge the gap between innovation and patient care.
Cliniexperts Research supports sponsors with expert regulatory guidance, structured monitoring, data management excellence, and cost-effective trial execution—making them a trusted CRO partner in the clinical research ecosystem.